Laboratory of Electrostatic Methods of Bioencapsulation
Head of Laboratory: Dorota Lewińska, PhD, DSc, Assoc. Prof.
Marcin Grzeczkowicz , PhD Eng.
Katarzyna Kramek-Romanowska, PhD Eng.
Adam Mirek, MSc Eng. (PhD student)
All elaborated methods of living cell bioencapsulation base on electrostatic microbead generator, exploiting electrostatic droplet formation principle and using impulse high voltages, allowing to manufacture spherical and uniform hydrogel microbeads of sizes from 0.2 to 3.0 mm of very narrow size distribution (variation coefficient of diameter below 10%).
The electrostatic droplet generator can work with:
• single nozzle (designed for microencapsulation of small amounts of cells – e.g. rat Langerhans islets)
• multi-nozzle head (useful for laboratory-scale microbead production)
• double-nozzle head (designed mainly for liquid-core microcapsule manufacturing- PATENT RP No 208384)
All types of the above devices have been tested in experiments with living cells (yeast cells – Fig. 1, WEHI-3B line cells, hepatic C3A and HepG2 line cells, rat Langerhans islets and human parathyroid cells – Fig. 2). It was concluded that applied electrostatic field is safe for the encapsulated living cells and does not cause any cell dysfunction.
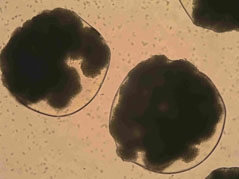
a) 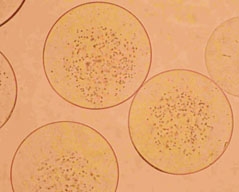 b)
b)
Fig. 1. Yeast cells encapsulated inside alginate microbeads by the double-nozzle technique:
a) just after manufacturing, b) after 48 h of the incubation.
Lewińska et al. Biocybernetics and Biomedical Engineering, 2008, 28(2), 69-84
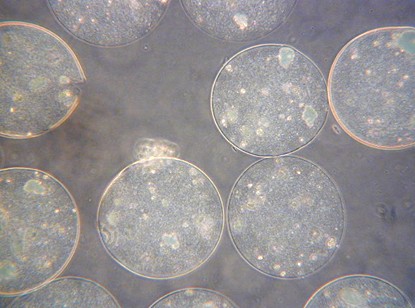
Fig. 2. Microencapsulated human parathyroid cells
• 3-nozzle head (PATENT RP No 208383)
Applying of a special 3-nozzle head (designed in our lab) enables one to obtain quite a new and unique type of alginate-polyethersulfone microcapsules in a one-step procedure. They consist of cell-friendly, hydrogel core covered by a semipermeable membrane made up of high molecular weight synthetic polymer. This kind of microcapsules (Fig. 3-5) could be wildly applied in medicine and biotechnology due to their improved mechanical and mass transport properties in comparison with classical APA (alginate-poly-lysine-alginate) or other polyelectrolyte microcapsules. Moreover, the one-step method gives a unique possibility to form thin (about 100 μm) membranes of different permeability using synthetic polymer mixtures of various chemical structure, physical and chemical properties as an affinity of membrane forming solution with matrix material is not necessary.
Fig. 3. The view of the alginate-polyethersulfone microcapsules Fig. 4. SEM image of the microcapsule interior.
Fig. 5. Optical images of the cross-sections of yeast-loaded alginate-polyethersulfone microcapsules: before (a) and after 24 h cultivation of yeast cells (b)
It has been also proved (Fig. 5) that the procedure does not destroy encapsulated yeast cells, which still possess proliferation ability. The applied polyethersulfone membrane is permeable for nutrients.
Keywords: microencapsulation of cells, electrostatic field, liquid-core microcapsules, alginate-polyethersulfone microcapsule, alginate microbeads
Selected publications:
• Lewińska D, Rosiński S, Weryński A: Influence of process conditions during impulsed electrostatic droplet formation on size distribution of hydrogel beads; Artif Cell, Blood Substitutes and Biotechnol, vol. 32, 1, 41-53, 2002
• Lewińska D, Rosiński S, Hunkeler D, Poncelet D, Weryński A: Mass transfer coefficient in characterization of gel beads and microcapsules, J. Membrane Sci, vol. 209(2): 533-540, 2002
• Rosiński S, Lewińska D, Migaj M, Woźniewicz B, Weryński A: Electrostatic microencapsulation of parathyroid cells as a tool for the investigation of cells activity after transplantation”; Landbauforschung Voelkenrode, Special Issue, 241, 47-50, 2002
• Hunkeler D, Wandrey Ch, Rosiński S, Lewińska D, Weryński A: Characterization of microcapsules (in: Fundamentals of Cell Immobilisation Biotechnology, eds. Nedovič V, Willaert R), Kluwer Academic Publishers, Dordrecht, Boston, London, 8A, 389-410, 2004
• Rosiński S, Lewińska D, Wójcik M, Orive G, Pedraz JL, Weryński A: Mass transfer characteristics of poly-lysine, poly-ornithine and poly-methylene-co-guanidine membrane coated alginate microcapsules. J Membrane Sci, vol. 254, 249-257, 2005
• Kinasiewicz A, Gautier A, Lewińska D, Bukowski J, Legallais C, Weryński A: Culture of C3A cells in alginate beads for fluidized bed bioartificial liver. Transplant Proc, vol. 39(9) 2911-13, 2007
• Lewińska D, Bukowski J, Kożuchowski M, Kinasiewicz A, Weryński A: Electrostatic microencapsulation of living cells. Biocyb and Biomed Eng, vol. 28(2), 69-84, 2008
• Prusse U, Bilancetti L, Bucko M, Bugarski B, Bukowski B, Gemainer P, Lewińska D, Massart B, Nastruzzi C, Nedovic V, Poncelet D, Siebenhaar S, Tobler L, Tosi A, Vikartovska A, Vorlop K-D: Comparison of different technologies for alginate bead production. Chem Papers, vol. 62(4), 364-374, 2008
• Kinasiewicz A, Gautier A, Lewińska D, Śmietanka A, Legallaise C, Weryński A: Three-dimensional growth of human hepatoma C3A cells within alginate beads for fluidized bioartificial liver. Int J Artif Organs, vol. 31(4), 340-347, 2008
• Kupikowska B, Lewińska D, Dudziński K, Jankowska-Śliwińska J, Grzeczkowicz M, Wojciechowski C: The influence of changes in composition of membrane-forming solution on the structure of alginate-polyethersulfone microcapsules. Biocyb and Biomed Eng, vol. 29(3), 61-69, 2009
• Lewińska D, Chwojnowski A, Wojciechowski C, Kupikowska-Stobba B, Grzeczkowicz M, Weryński A: Electrostatic droplet generator with 3-coaxial-nozzle head for microencapsulation of living cells in hydrogel covered by synthetic polymer membranes. Sep Sci and Technol, vol. 47(3), 463-469, 2012
• Chwojnowski A, Wojciechowski C, Lewińska D, Łukowska E, Nowak J, Kupikowska-Stobba B, Grzeczkowicz M.: Studies on the structure of semi-permeable membranes by means of SEM. Problems and potential sources of errors. Biocybernetics and Biomedical Engineering, vol. 32(1), 51-64, 2012
• Kupikowska-Stobba B, Lewińska D, Grzeczkowicz M: Chemical method for retrieval of cells encapsulated in alginate-polyethersulfone microcapsules: Artif Cells Nanomed and Biotechno, vol. 42(3), 151-160, 2014, DOI:10.3109/21691401.2013.800083
• Przytulska M, Kulikowski J.L., Lewińska D, Grzeczkowicz, Kupikowska-Stobba B: Computer-Aided Image Analysis for Microcapsules’ Quality Assessment. Biocyb and Biomed Eng, vol. 35(4), 342-350, 2015, DOI:10.1016/j.bbe.2015.05.005
• Lewińska D, Grzeczkowicz M, Kupikowska-Stobba B: Influence of electric parameters on the alginate-polyethersulfone microcapsule structure. Desalination and Water Treatment vol. 64, 400-408, 2017 DOI: 10.5004/dwt.2017.11407. 64 (2017)
• Korycka, P., Mirek, A., Kramek-Romanowska, K., Grzeczkowicz, M., Lewińska, D., Effect of electrospinning process variables on the size of polymer fibers and bead-on-string structures established with a 23 factorial design. Beilstein J. Nanotechnol. 9, 2466–24780, 2018, DOI:10.3762/bjnano.9.2312466–2478
Patents:
• Lewińska D, Bukowski J, Rosiński S, Kożuchowski M, Weryński A: One-step method of microcapsule preparation, especially with entrapped living cells, cell aggregates or bioactive substances and the one-step device for this purpose. PATENT RP No 208384, 2004
• Lewińska D, Chwojnowski A, Jankowska-Śliwińska J, Weryński A: Microcapsules containing biologically active substances, especially living cells and/or microorganisms, alternatively genetically modified or natural or synthetic for biomedical applications, the way of their manufacturing and the device for this purpose. PATENT RP No 208383, 2007
• Lewińska D, Kupikowska-Stobba B, Grzeczkowicz M, Chwojnowski A, Łukowska E: Method for quantitative determination of cell concentration. PATENT RP 223717, 2012
Patent Pendings:
• Chwojnowski A, Łukowska E, Wojciechowski C, Lewińska D: Semi-permeable membrane and a method for the preparation of the membrane. Polish Patent Pending No P.403657 of 24.04.2013
• Kutkowska A, Lewińska D, Grzeczkowicz M, Chwojnowski A: Method and set-up for forming spherical microbeads in slow-gelling systems. Polish Patent Pending No P.417705 of 23.06.2016
International cooperation:
• 1999 - 2004 direct scientific cooperation within the frame of the European Project COST 840 Action “Bioencapsulation. Innovations and Technologies” with research groups from:
- Thüne Insitute of Agricultural Technology, Braunschweig, Germany;
- Institute of Chemistry Slovak Academy of Sciences, Bratislava, Slovakia;
- Deptartment of Chemical Engineering Faculty of Technology and Metallurgy, Belgrade, Serbia
and Montenegro;
- ENITIAA, UMR CNRS GEPEA, Nantes, France
- Laboratory of Polyelectrolytes and Biomacromolecules, Swiss Federal Institute
of Technology, Lausanne, Switzerland;
- Institute of Pharmaceutical Technology and Biopharmaceutics, University of Vienna, Austria;
- School of Chemical Engineering, University of Birmingham, Edgbaston, Birmingham, Great
Britain;
- Laboratory of Chemical and Biological Engineering, Swiss Federal Institute of Technology,
Lausanne, Switzerland;;
- Department of Pharmacy and Pharmaceutical Technology, University of Basque Country, Vitoria,
Spain.
• 2005-2007 cooperation with group led by prof. Cecile Legallais from the University of Technology, Compiegne, CNRS, UMR 6600, Compiegne within the bilateral project Polonium on the subject: “Modification of alginate beads with hepatocytes for bioartificial liver.”
Grants:
• “COST 840 Action – Selection of conditions in the process of microcapsule formation using an electrostatic field and coating microcapsules in membrane, and development of a method for studying their transport properties in vitro.” Special Grant SPUB-M No. 56/E-83/ SPUB/COST/P-04/DZ 62/99 funded by Polish State Committee for Scientific Research realized between 1998-2001
• “A new therapeutic strategy in transplantology. Allotransplantation of human parathyroid cells in vitro in therapy of hypoparathyroidism without immunosuppression” Research project PB 538/PO5/98/15 funded by Polish State Committee for Scientific Research realized between 1998-2001
• “Developing and studying a modified electrostatic microcapsule generator. Performing transport studies of microcapsules produced within the COST 840 project” Special Grant SPUB-M No. 56/E-83/ SPUB/COST/P-04/DZ584/2002-2004 funded by Polish State Committee for Scientific Research realized between 2002-2004
• “The elaboration of the method of structural homogenous membrane preparation in alginate-polyethersulfone microcapsule applying the computer-aided image analysis methods.” 29-month grant No 4151/B/T02/2009/36 funded by Polish State Committee for Scientific Research 2009 - 2011.
MENU
- News
- About the Institute
- Mission & Authorities
- HR Excellence
- Doctoral Schools
- Doctoral Studies
- Academic degrees
- Scientific activity
- Institute offers
- ICB
- Publishing
- Library
- Conference Centre
- Useful Links
- Public procurement
- Employees
- Gallery
- For Media
- Contact
- Privacy policy
Nałęcz Institute of Biocybernetics and Biomedical Engineering PAS, Ks. Trojdena 4 st., 02-109 Warsaw, POLAND
E-mail:This email address is being protected from spambots. You need JavaScript enabled to view it.; Phone: (+48) 22 592 59 00;
Copyright(c) 2016 IBBE PAS
All rights reserved
In order to provide you with the best online experience this website uses cookies. Delete cookies
In order to provide you with the best online experience this website uses cookies.
By using our website, you agree to our use of cookies. Learn more
Useful Links
Privacy policy
Ministry of Science and Higher Education
Polish Science Database (Nauka Polska)
Department of Patents - website in Polish
Sholarship granting institutions
The National Centre for Research and Development
The Polish National Agency for Academic Exchange
European Union Funding
Publication databases
ISI Web of Knowledge Journal Citation Report
BazTech - website in Polish
ICM - Virtual Library of Science - website in Polish
Other
Datasets